Blood-based molecular profiling reagents are moving into the spotlight as precision oncology testing continues to shift toward minimally invasive sample types. On May 8, 2026, Caris Life Sciences announced that it had submitted an application to the New York State Department of Health Clinical Laboratory Evaluation Program seeking authorization to perform Caris Assure, its blood-based molecular profiling test, on specimens originating from New York State. For diagnostic developers, the news is a useful reminder: liquid biopsy workflows depend not only on sequencing platforms and bioinformatics, but also on consistent raw materials, sample preparation chemistry, enzyme performance and documented quality control.
This article uses that news as a practical starting point to review reagent strategy for blood-based molecular profiling diagnostics, especially where cfDNA/cfRNA handling, NGS library preparation, qPCR verification and OEM scale-up must work together under a regulated quality mindset.
Why blood-based molecular profiling reagents matter now
Blood-based molecular profiling can help laboratories analyze tumor-associated biomarkers from a minimally invasive blood sample. In practice, however, the analytical signal can be limited by low nucleic acid input, fragmented templates, sample transport conditions, inhibitors, batch variation and background noise. These variables make reagent selection a strategic issue rather than a routine purchasing decision.
Several current market signals point in the same direction:
- Oncology testing companies are expanding blood-based molecular profiling and MRD-related menus.
- Clinical laboratories increasingly need workflows that combine extraction, library preparation, amplification, sequencing and orthogonal QC.
- Regulated or near-regulated diagnostic programs require supplier documentation, lot traceability and change-control discipline.
- Global IVD developers need reagent formats that can support automation, OEM transfer and stable supply.
For reagent and assay development teams, the key question is: which raw material attributes reduce workflow risk before the assay reaches verification, validation or routine manufacturing?
Critical workflow stages for blood-based profiling assays
A typical liquid biopsy or blood-based profiling workflow may include plasma separation, nucleic acid extraction, quality assessment, library preparation, target enrichment or amplification, sequencing, data analysis and result confirmation. The reagent strategy should be planned around the weakest points of the workflow.
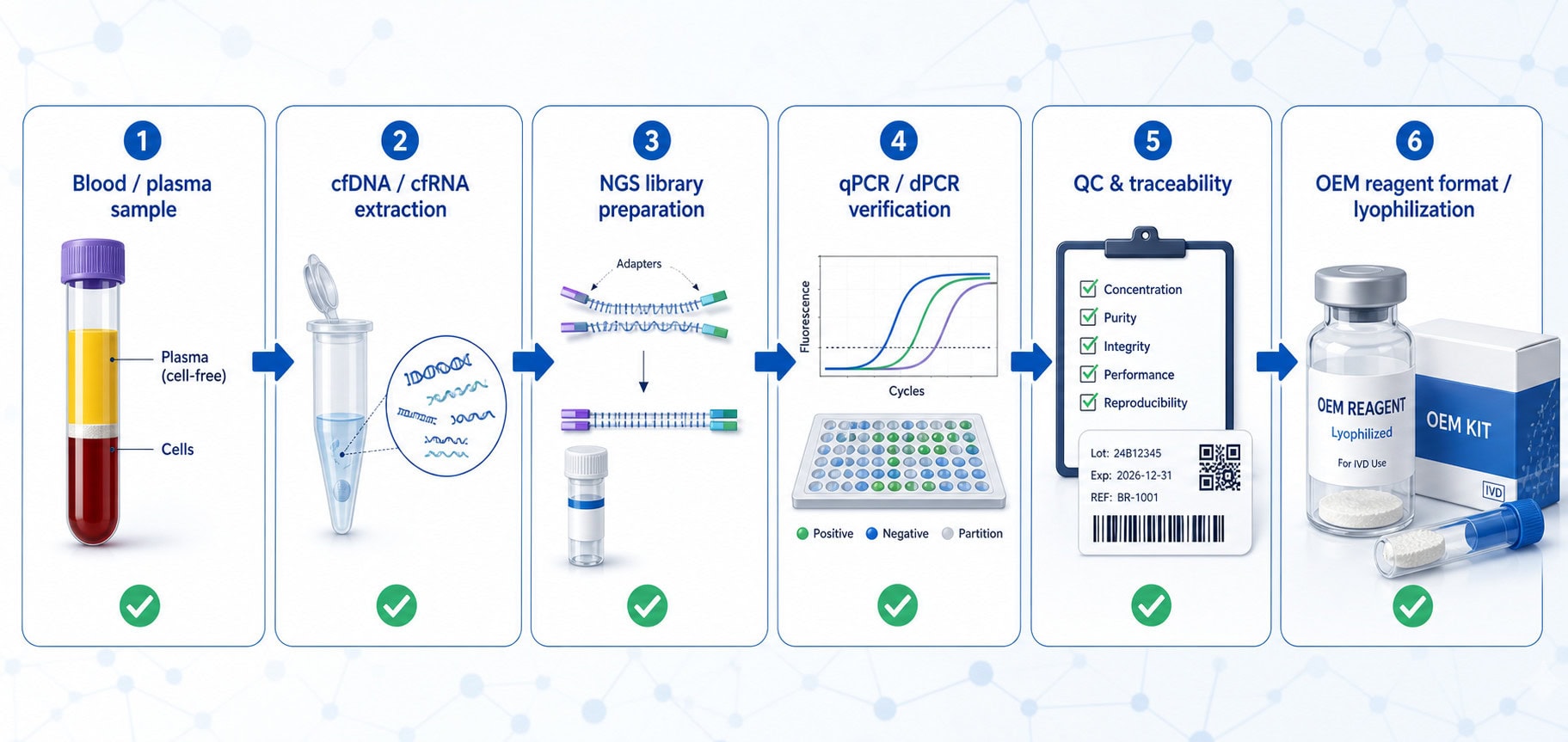
1. cfDNA and cfRNA extraction
Extraction chemistry must preserve short nucleic acid fragments while minimizing inhibitors and carryover contaminants. For cfDNA workflows, recovery of low-input fragmented DNA is especially important. For RNA-related profiling, RNase control and process timing become even more critical.
2. Enzymatic steps in NGS library preparation
Library preparation often depends on high-performance enzymes such as polymerases, ligases, end repair enzymes, reverse transcriptases or structure-specific nucleases. Small shifts in activity, purity or buffer compatibility can affect library yield, insert-size distribution, duplication rate and downstream sequencing quality. Biori’s NGS Selection page provides a relevant entry point for teams evaluating enzyme and reagent options for genomics workflows.
3. qPCR or digital PCR verification
Many assay development teams use qPCR, RT-qPCR or dPCR as orthogonal tools for input QC, copy number assessment, contamination monitoring or specific variant verification. Polymerase robustness, inhibitor tolerance, UNG/dUTP carryover control and multiplex compatibility can all influence confidence in verification data. For PCR-related raw materials, see Biori’s Molecular Diagnostics Selection resources.
4. Manufacturing transfer and OEM formats
Once an assay progresses beyond feasibility, reagent format becomes a business and quality decision. Diagnostic teams may need glycerol-free enzymes, lyophilized master mixes, automated filling, custom concentration, private-label documentation, packaging support or quality release files. Biori offers OEM and custom manufacturing services and lyophilization service for molecular diagnostics for teams planning a manufacturable reagent supply chain.
Reagent attributes to evaluate before assay lock
Blood-based molecular profiling assays should evaluate reagents across performance, formulation and quality dimensions. The table below summarizes practical checkpoints.
| Workflow area | Key reagent question | Why it matters |
|---|---|---|
| Extraction | Does the chemistry recover short, low-input nucleic acid fragments consistently? | Low recovery can reduce sensitivity before amplification or sequencing begins. |
| Library preparation | Are enzyme activity, purity and buffer compatibility stable lot to lot? | Variation may affect library yield, bias and reproducibility. |
| Amplification/QC | Can qPCR reagents tolerate inhibitors and support multiplex targets? | Verification assays often operate with limited clinical sample material. |
| Formulation | Can the reagent be supplied in liquid, glycerol-free or lyophilized formats? | Format affects cold-chain burden, automation, transport and kit assembly. |
| Documentation | Are CoA, traceability, QC release and change-control data available? | Documentation supports regulated development and supplier qualification. |
Quality control considerations for diagnostic developers
As blood-based molecular profiling programs move closer to clinical use, teams should build a reagent QC plan that matches the intended assay risk. Useful checkpoints include:
- Incoming raw material qualification: review CoA, activity definition, storage conditions, impurity profile and supplier quality system information.
- Functional QC: test reagents in the actual assay matrix rather than relying only on generic activity units.
- Lot bridging: compare new lots against reference lots using clinically relevant sample types or contrived low-input materials.
- Stability monitoring: evaluate freeze-thaw, shipping stress, accelerated stability and open-vial stability where applicable.
- Contamination control: monitor nuclease, carryover amplicon and microbial risks according to the assay context.
- Manufacturing documentation: maintain traceability, batch records and change notification expectations before scale-up.
For broader quality-system context, Biori’s Manufacturing & Quality page outlines ISO 9001 / ISO 13485 / GMP-oriented capabilities relevant to reagent development and manufacturing programs.
How Biori can support blood-based molecular profiling reagent programs
Biori supports international customers developing molecular diagnostic and biopharma workflows with enzyme raw materials, PCR/RT-qPCR reagents, NGS-related solutions, OEM/custom manufacturing, lyophilized and glycerol-free formulation options, and quality documentation support. For blood-based molecular profiling programs, this can include discussion around:
- DNA polymerases, hot-start PCR enzymes and qPCR master mix components for verification assays.
- NGS workflow enzymes and customized reagent development for sample preparation and library workflows.
- OEM formulation, filling, packaging and documentation for IVD reagent programs.
- Lyophilized or glycerol-free formats for transport stability and kit integration.
- HCD/HCP and process impurity testing considerations for biopharma or advanced therapy contexts through QA/QC product selection and HCD/HCP custom assay development.
For adjacent RNA and therapeutic manufacturing programs, Biori also provides a complete portfolio of GMP-grade IVT raw materials for mRNA manufacturing, including key enzymes and workflow reagents such as T7 RNA polymerase, capping-related enzymes, DNase, RNase inhibitor and supporting customization/OEM services. This capability is relevant when diagnostic, vaccine or RNA platform teams require higher-grade raw material planning across multiple nucleic acid workflows.
FAQ: blood-based molecular profiling reagent strategy
What are blood-based molecular profiling reagents?
They are extraction chemistries, enzymes, buffers, master mixes, library preparation components and QC reagents used to analyze molecular biomarkers from blood-derived samples such as plasma, cfDNA or cfRNA.
Why is low-input performance important for cfDNA profiling?
Blood-derived tumor DNA can be fragmented and present at low abundance. Reagents must support efficient recovery, conversion and amplification without adding avoidable bias or inhibition.
When should diagnostic teams consider OEM reagent development?
OEM development is useful when a workflow needs custom concentration, private-label supply, lyophilized format, documentation, lot consistency or scale-up support beyond standard catalog reagents.
Can qPCR still be useful in NGS-based profiling workflows?
Yes. qPCR or RT-qPCR can support sample input checks, contamination monitoring, orthogonal verification, assay development and process troubleshooting even when sequencing is the primary readout.
References
- Caris Life Sciences: application to NYSDOH for Caris Assure blood-based testing authorization
- New York State Clinical Laboratory Evaluation Program (CLEP), Wadsworth Center
Discuss your reagent strategy with Biori
If you are developing blood-based molecular profiling, cfDNA testing, NGS sample preparation, qPCR verification or OEM diagnostic reagents, contact Biori to discuss enzyme selection, formulation options, lyophilization, glycerol-free supply, quality documentation and custom manufacturing support for overseas programs.
