NGS sample preparation is becoming a more important part of molecular diagnostics as sequencing-based assays move from research settings toward clinical, infectious disease, oncology, and microbial detection workflows. Recent industry coverage of new tools for microbial detection and NGS, together with continued activity in NGS-based companion diagnostics, highlights a practical point for assay developers: the performance of a sequencing test depends heavily on what happens before the sequencing run.
For IVD developers and molecular diagnostic manufacturers, sample preparation is not only a laboratory step. It is a reagent strategy that connects nucleic acid extraction, enzymatic library preparation, amplification, QC, documentation, and scalable supply.
Why NGS Sample Preparation Matters in Molecular Diagnostics
NGS-based assays often need to handle low-input, degraded, or inhibitor-rich samples. Depending on the application, the sample may come from blood, FFPE tissue, respiratory swabs, microbial cultures, plasma, or other complex matrices. Each sample type can influence extraction efficiency, library complexity, bias, and downstream data quality.
When diagnostic workflows require consistent results across sites, batches, and operators, upstream reagent design becomes as important as sequencing capacity. A robust NGS sample preparation workflow helps reduce variability before it becomes an expensive sequencing or bioinformatics problem.
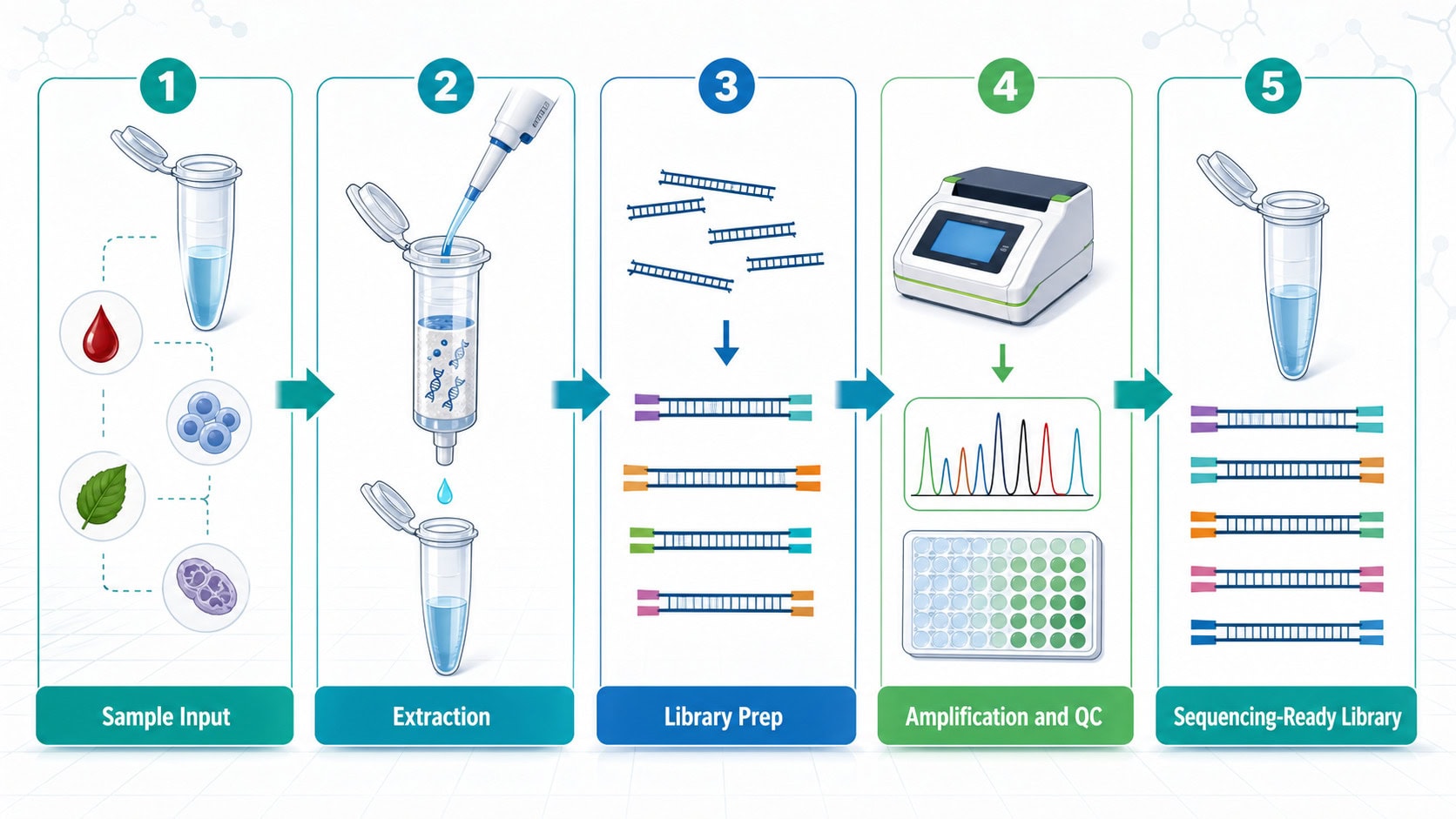
NGS Sample Preparation Workflow: Key Reagent Checkpoints
A practical NGS sample preparation workflow usually includes several reagent-sensitive steps. Each step can affect library yield, uniformity, contamination risk, and reproducibility.
1. Nucleic acid extraction and input quality
Extraction chemistry must be matched to the sample type and assay goal. For molecular diagnostics, developers should evaluate recovery, inhibitor carryover, sample-to-sample consistency, and compatibility with downstream enzymatic steps.
2. Fragmentation or end-repair strategy
Library preparation may require fragmentation, end repair, A-tailing, or other enzymatic preparation steps. Enzyme performance, buffer compatibility, and reaction robustness can influence insert-size distribution and final library complexity.
3. Adapter ligation and amplification
Adapter ligation efficiency and amplification bias are major sources of variability in NGS assays. Ligases, polymerases, master mixes, and cycle conditions should be evaluated for sensitivity, specificity, and reproducibility across expected sample inputs.
4. Library QC and release criteria
Before sequencing, diagnostic teams often need QC checkpoints such as library concentration, fragment distribution, contamination controls, and assay-specific acceptance criteria. These checkpoints help prevent poor-quality libraries from consuming sequencing capacity.
How to Choose NGS Library Prep Reagents for Diagnostic Development
For molecular diagnostic applications, reagent selection should be based on more than catalog performance. Teams should evaluate whether the supplier can support scale-up, documentation, quality-system expectations, and format customization.
| Selection area | Why it matters | Supplier question |
|---|---|---|
| Enzyme consistency | Supports reproducible library yield and size distribution | Can lot-to-lot performance data be provided? |
| Matrix tolerance | Reduces failure risk with complex clinical samples | Has the reagent been tested with relevant sample types? |
| Workflow format | Improves usability in kit development and manufacturing | Are premix, lyophilized, or glycerol-free formats available? |
| OEM readiness | Supports commercial IVD or RUO kit development | Can the supplier support private-label or customized reagent supply? |
| Quality documentation | Supports regulated development and customer audits | What ISO/GMP-related documentation can be provided? |
Avoiding Common NGS Sample Preparation Pitfalls
- Optimizing sequencing before optimizing sample prep: poor extraction or biased libraries cannot always be fixed downstream.
- Ignoring reagent format early: a liquid R&D workflow may not translate smoothly into a stable diagnostic kit format.
- Using generic enzymes without supply planning: diagnostic developers should consider lot consistency, documentation, and long-term availability.
- Separating workflow design from QC design: sample preparation, library QC, and final assay acceptance criteria should be developed together.
Where OEM Support Adds Value for NGS Diagnostic Kits
As NGS-based assays move closer to routine diagnostic use, developers often need reagents that can be customized for performance, format, packaging, and documentation. OEM support can help convert a laboratory protocol into a more manufacturable kit workflow.
Useful OEM options may include:
- Customized enzymes for library preparation and amplification
- Premixed reagent formats to reduce operator variability
- Lyophilized or glycerol-free formulations for stability and logistics
- Matched extraction and amplification components
- Private-label supply for RUO or IVD-oriented kit developers
- Documentation support aligned with ISO 13485, ISO 9001, and GMP-related expectations
How Biori Supports NGS and Molecular Diagnostic Workflows
Biori supports international customers with molecular diagnostic raw materials, PCR/RT-qPCR reagents, NGS-related enzyme and reagent solutions, nucleic acid workflow components, and OEM/customization services. For NGS sample preparation, Biori can help developers evaluate enzyme performance, reagent format, workflow compatibility, and scalable supply strategy.
For adjacent biomanufacturing and mRNA workflows, Biori also provides a complete portfolio of GMP-grade IVT raw materials for mRNA manufacturing, including key enzymes and workflow reagents, with OEM and customization support.
CTA: If you are developing NGS-based molecular diagnostics, sequencing sample preparation kits, or customized reagent workflows, contact Biori to discuss NGS library prep reagents, PCR/qPCR components, lyophilized or glycerol-free formats, and OEM manufacturing support.
- Explore Biori NGS library prep and sequencing reagent solutions
- Learn about Biori OEM and custom manufacturing services
- View Biori PCR/qPCR and molecular diagnostics reagent solutions
FAQ: NGS Sample Preparation for Molecular Diagnostics
Why is sample preparation critical for NGS diagnostics?
Sample preparation determines how much usable nucleic acid enters the library preparation process and how consistently libraries are generated across sample types and batches.
What enzymes are important in NGS library preparation?
Depending on the workflow, key enzymes may include polymerases, ligases, reverse transcriptases, fragmentation or end-repair enzymes, and amplification master mix components.
Can NGS sample preparation reagents be customized for OEM kits?
Yes. OEM customization can include enzyme selection, premix formats, lyophilization, glycerol-free formulations, packaging, labeling, and documentation support.
How should diagnostic teams evaluate an NGS reagent supplier?
Teams should consider performance data, lot consistency, format flexibility, quality-system documentation, technical support, and ability to support scale-up or private-label supply.
References
- Technology Networks. Recent coverage on new tools advancing microbial detection and NGS, April 2026.
- BioSpace. Foundation Medicine and Bristol Myers Squibb collaboration coverage related to NGS companion diagnostics, April 2026.
