The recent collaboration between Foundation Medicine and Fulgent Genetics to offer a new gene test for drug metabolism underscores a broader trend: pharmacogenomic testing reagent strategy is becoming more important as clinical laboratories translate genomic information into routine diagnostic workflows.
For diagnostic developers, the opportunity is not only the test menu itself. It is the ability to build reliable sample preparation, amplification, sequencing or genotyping, controls, QC release and documentation systems that can scale from validation to routine clinical use.
Why pharmacogenomic testing reagent strategy matters now
Pharmacogenomic assays often evaluate variants in genes linked to drug metabolism, transport or response. Depending on the panel design, laboratories may use targeted PCR, qPCR, Sanger sequencing, NGS panels, or hybrid workflows. Each format places different demands on enzymes, buffers, primers, probes, controls and documentation.
Recent industry news around new clinical gene testing services suggests continued interest in bringing genomic information closer to prescribing decisions. For reagent and assay teams, this creates practical questions: how should DNA extraction be controlled, which amplification chemistry is robust enough, and how should lot-to-lot performance be documented for regulated customers?
Core reagent considerations for clinical pharmacogenomic assays
1. DNA extraction and input quality
Pharmacogenomic testing typically starts with genomic DNA from blood, buccal swabs or other validated sample types. Extraction reagents should deliver consistent yield, purity and inhibitor tolerance across sample matrices. For OEM programs, developers should define acceptable input ranges, carryover risks, storage conditions and control materials early in development.
2. PCR, qPCR and NGS preparation enzymes
Targeted pharmacogenomic assays may rely on hot-start DNA polymerases, multiplex PCR systems, qPCR master mixes, reverse-complement primer design controls, ligases, fragmentation enzymes or library-preparation reagents. The selection criteria should include specificity, uniformity across targets, inhibitor tolerance, thermal stability and compatibility with automation.
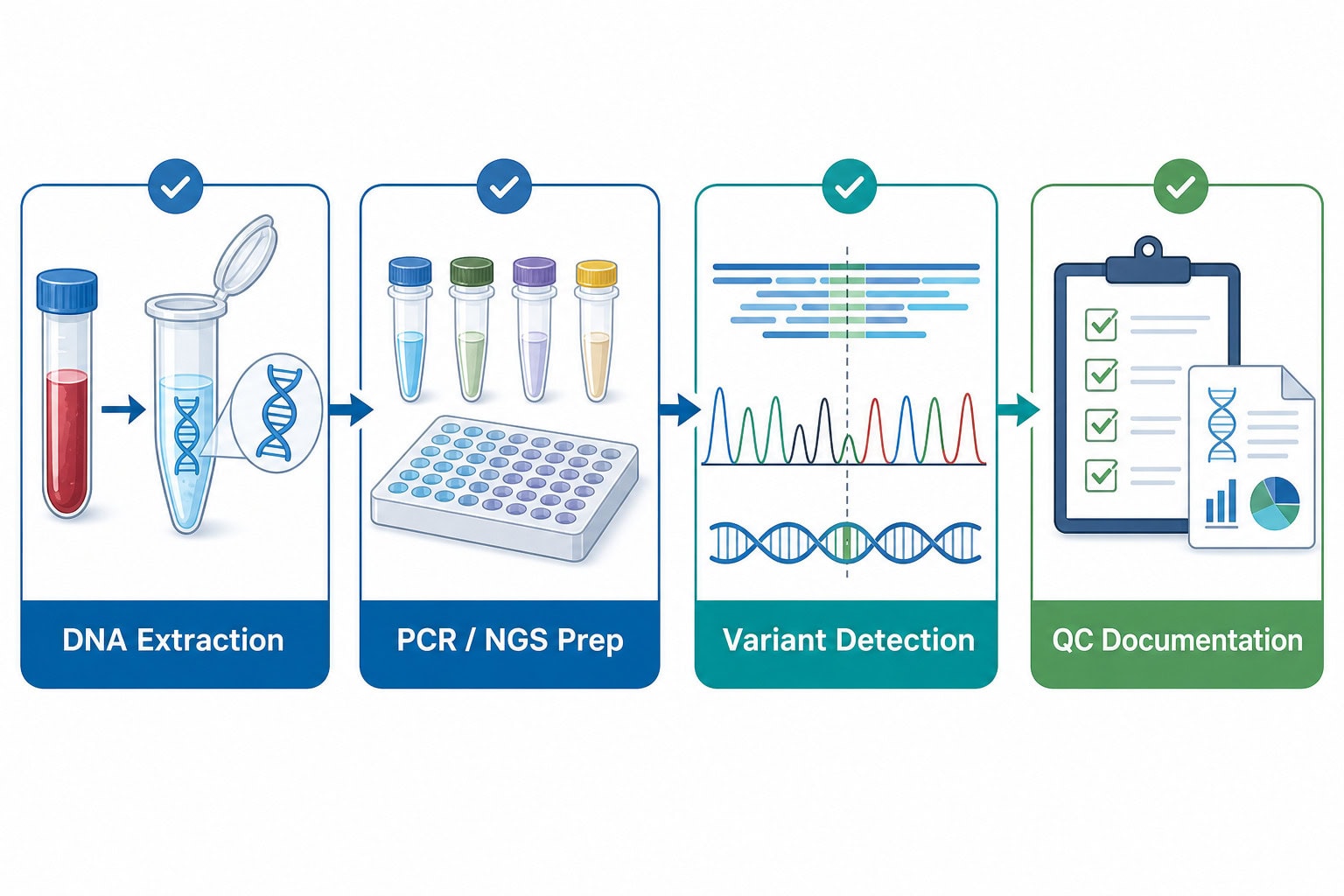
3. Controls, reference materials and contamination management
Because pharmacogenomic results can affect therapy decisions, assay teams need clear positive, negative and no-template control strategies. Laboratories should evaluate cross-contamination prevention, amplicon carryover controls, sample tracking and acceptance criteria for each batch.
4. QC documentation and supplier traceability
Clinical diagnostic customers increasingly expect more than reagent performance claims. They need Certificates of Analysis, lot traceability, change-control communication, stability data, and quality-system documentation aligned with their validation and regulatory pathways. Biori’s manufacturing and quality capabilities can support programs requiring consistent supply and documentation.
How OEM reagent programs can reduce development risk
For laboratories and diagnostic manufacturers, an OEM reagent strategy can shorten the path from assay concept to stable routine supply. Key development steps include:
- Feasibility testing: compare enzyme systems, buffers and cycling conditions across representative genomic targets.
- Formulation optimization: adjust salts, Mg2+, stabilizers and additives for sample type and platform compatibility.
- Stability planning: evaluate liquid, glycerol-free and lyophilized formats for storage and shipping requirements.
- Manufacturing scale-up: define batch size, release tests, packaging, labeling and documentation expectations.
- Lifecycle support: manage lot changes, performance monitoring and customer-specific documentation.
For teams building molecular diagnostic assays, Biori provides molecular diagnostics raw materials, NGS-related enzyme and reagent options, and OEM and custom manufacturing services for assay-ready formats.
When lyophilized or glycerol-free formats are useful
Pharmacogenomic tests may be deployed across centralized laboratories, regional networks or partner sites. In these settings, lyophilized or glycerol-free reagent formats can simplify cold-chain requirements, reduce pipetting complexity and support more consistent handling. Biori’s lyophilization service for molecular diagnostics can help teams evaluate bead, cake or premix formats for PCR and RT-qPCR workflows.
Connecting pharmacogenomics with broader biomanufacturing QC
Although pharmacogenomic testing is primarily a clinical diagnostics application, many organizations operate across diagnostics, biologics and advanced therapy development. The same quality mindset—traceable reagents, validated workflows and well-documented release criteria—also applies to residual Host Cell DNA, Host Cell Protein and other biopharma QC assays. Biori supports HCD/HCP and QA/QC product selection as well as HCD/HCP custom assay development.
For adjacent mRNA and IVT programs, Biori also provides a complete portfolio of GMP-grade IVT raw materials for mRNA manufacturing, including key enzymes and workflow reagents, with OEM/customization support.
FAQ: pharmacogenomic testing reagent strategy
What is a pharmacogenomic testing reagent strategy?
It is the plan for selecting, validating and supplying the extraction reagents, amplification enzymes, NGS or qPCR reagents, controls, stability formats and QC documentation needed for pharmacogenomic assays.
Which reagent formats are common in pharmacogenomic testing?
Common formats include DNA extraction reagents, PCR/qPCR master mixes, primers and probes, library-preparation reagents, sequencing enzymes, control materials, and sometimes lyophilized or glycerol-free assay-ready mixes.
Why is OEM support important for clinical diagnostic reagent programs?
OEM support can help diagnostic developers move from feasibility to routine supply by combining formulation optimization, batch manufacturing, QC release, packaging, documentation and change-control support.
Can lyophilized reagents support pharmacogenomic workflows?
Yes, when properly developed and validated, lyophilized formats may improve handling consistency, shipping flexibility and workflow simplicity for PCR or qPCR-based pharmacogenomic assays.
References
- Fierce Biotech: Foundation Medicine links with Fulgent Genetics to offer new gene test
- FDA: Table of Pharmacogenetic Associations
Discuss OEM pharmacogenomic reagent development with Biori
If you are developing pharmacogenomic, PCR/qPCR, NGS or broader molecular diagnostic workflows, Biori can support enzyme selection, formulation optimization, lyophilized or glycerol-free formats, OEM manufacturing and QC documentation. Contact Biori to discuss your overseas diagnostic reagent program.
